Antibody conjugation
This protocol outlines antibody conjugation by NHS-esters.
🚛 Reagents needed 📦 :
| Item | Item article number | Distributor | Vendor |
|---|---|---|---|
| Anhydrous DMSO | 440140 | Merck | SigmaAldrich |
| UltraPure™ DNase/RNase-Free Distilled Water | 10977035 | ThermoFisher | Invitrogen |
| 0.5 M NaHCO3, pH 8 | |||
| NHS-ester or similar, here we use TCO-PEG4-TFP Ester |
CCT-1398 |
BioNordika | |
| Zeba Spin Desalting Columns, 0.5 ml,40K MWCO |
11796436 | ThermoFisher | ThermoFisher |
| Amicon ultra 30K 0.5mL (UFC5030BK) | UFC5030BK |
Merck | Amicon |
| Tris (1 M), pH 8.0, RNase-free | AM9855G | ThermoFisher | Invitrogen |
Protocol
⚗️Antibody Purification.
BSA, azide, or glycine that are often added by the manufacturer to the antibody, this step is to remove such reagents from the antibody buffer.
Amine-containing compounds (glycine, Tris, or ammonium ions) and stabilising proteins (BSA) will interfere with the NHS to amino labelling.
1️⃣ Add 500 ul of 1X PBS to moisten the cellulose membrane of the Amicon ultra 10K 0.5mL (UFC501096).
2️⃣ Centrifuge at 14,000 g for 3 min and the discard both the flow-through and the PBS at the bottom of the device.
3️⃣Wash the antibody in 1X PBS by taking 80 ul of antibody at 2 mg/mL and fill up with 500 µL of 1X PBS.
Add desired antibody and amount together with 500 µL of 1X PBS and centrifuge for 10 min @4000 g.
4️⃣ Discard the flow-through and add 500 µL of 1X PBS and centrifuge again for 10 min @14,000 g.
5️⃣ Collect purified antibody from the device by reverse spin at 1000 g for 2 min.
6️⃣Measure the absorbance of the antibody with a NanoDrop at 280 nm (A280). Write down the concentration (mg/mL) of the antibody. This number will later be used to calculate Degree of Labeling (DOL) as well as yield.
⚗️NHS preparation.
1️⃣Take out the NHS ester from the desiccator in the fridge and place within a inert gas seal if you intend to use it again. Importantly let it thaw completely for 20 min before opening the vial since otherwise moisture will condensate on the walls of the vial and will immediately induce hydrolysis of the ester.
Dissolve amine-reactive compound (NHS ester) in DMSO to 10 mg/mL (i.e. 100 µL of DMSO is added to 1 mg compound).
⚗️Antibody Labeling.
Prepare the unlabeled antibody stock to 2 mg/mL. Have it diluted in 0.5 M carbonate/bicarbonate (pH 8.75) buffer and aim to have a ~100 mM carbonate/bicarbonate concentration.
Here we will label the antibody at 3 molar rations 1:15, 1:9, and 1:3. The ester always needs to be in molar excess to the antibody.
1️⃣ Calculate the amount of NHS ester in DMSO you need to add to the antibody. The antibody is a Goat anti-rabbit in this example with a molecular weight of 144 kDa and we will label 160 uL in each reaction tube:
$15 \times \left( \frac{0.16 \ \text{mL } \times \ 2 \text{ mg/mL Antibody}}{144000 \ \text{Da Antibody weight}} \right) = 0.0000333 \ \text{mmol ester}$
$9 \times \left( \frac{0.16 \ \text{mL } \times \ 2 \text{ mg/mL Antibody}}{144000 \ \text{Da Antibody weight}} \right) = 0.00002 \ \text{mmol ester}$
$3 \times \left( \frac{0.16 \ \text{mL } \times \ 2 \text{ mg/mL Antibody}}{144000 \ \text{Da Antibody weight}} \right) = 0.00000666 \ \text{mmol ester}$
Then calculate how much volume needs to be added to the 160 uL antibody TCO-PEG4-TFP here has a molecular weight of 565.56 g/mol and we have the stock in DMSO at 1 mg in 100 uL DMSO (100 uL / 1 mg):
1:15: $0.0000333 \text{ mmol ester} \times 565.56 \text{ g/mol} \ \times \left( \frac{100 \text{ uL DMSO}}{1 \text{ mg Ester}} \right) = 1.885577 \text{ uL} \sim 1.9 \text{ uL}$
1:9: $0.00002 \text{ mmol ester} \times 565.56 \text{ g/mol} \ \times \left( \frac{100 \text{ uL DMSO}}{1 \text{ mg Ester}} \right) = 1.13112 \text{ uL} \sim 1.13 \text{ uL}$
1:3: $0.00000666 \text{ mmol ester} \times 565.56 \text{ g/mol} \ \times \left( \frac{100 \text{ uL DMSO}}{1 \text{ mg Ester}} \right) = 0.376663 \text{ uL} \sim 0.38 \text{ uL}$
All of these calculations can be done for any custom antibody or ester using the forms below:
Concentrations to evaluate (antibody:ester)
Antibody Settings
Ester Settings
2️⃣ Add the NHS to the antibodies in a tube. Pipette mix 10 times.
3️⃣ Incubate 1h in Room Temp in the dark 🌌, or for 2h on ice.
4️⃣ Quench the reaction by adding 1M Tris to a final concentration of 100 mM.
Tris contains a primary amine and thus will quench the unreacted NHS groups.
| Reagent | Volume | Final concentration |
|---|---|---|
| Antibody conjugation mixture | 160 µl | NA |
| Nuclease-free water | 20 µl | NA |
| Tris 1M pH 8.0 | 20 µl | 100 mM |
| TOTAL: | 200 µl |
❄️ Incubate 15 min on ice 🧊.
5️⃣Equilibrating Zeba spin columns:
- Spin 1,500 g for 1 min at room temp. Discard the elute.
- Add 300 μl of 1X PBS. Spin 1,500 g for 1 min at room temp. Discard the elute.
- Repeat one more rince with 300 μl of 1X PBS and discard the elute.
6️⃣Filter through Zeba:
- Add the volume of the antibody conjugate (200 μl) and let it absorb.
- At the end add some 15 μl 1xPBS to the column.
- Spin 1,500 g for 2 min at room temp.
7️⃣ Wash away excess NHS and concentrate further by Amicon ultra 30K 0.5mL (UFC5030BK) centrifugation using 500 ul of 1xPBS.
- Spin the device at 14,000 × g for 10 - 30 min for concentrating. 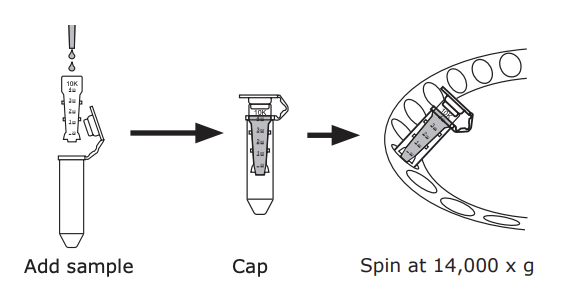
- Reverse spin at 1,000 × g for 2 min.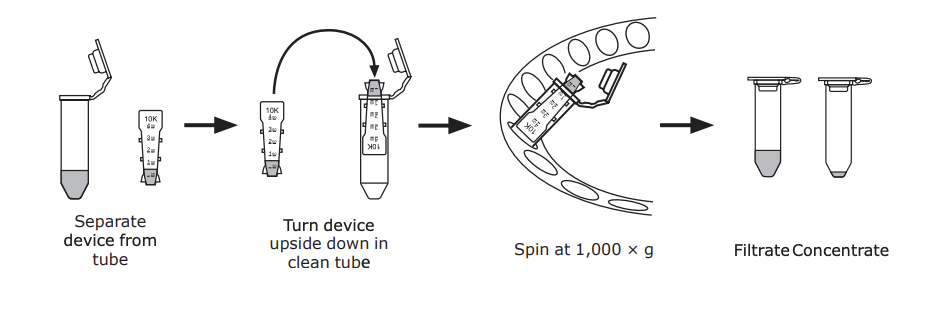
8️⃣Measure on NanoDrop and compare to your reference (⚗️Antibody Purification point 6️⃣). Dilute with 1xPBS to 2 mg/mL stock.